
A “carb” is short for carbohydrate. If you take a look at the two parts of that word, you’ll get a big hint as to how complex carbs are broken down by the body and how complex carbs are built in the first place! The term “carbo” refers to carbon, one of the most basic and essential atoms to all living things. In fact, when we say we are “organic life forms,” what we’re really saying is that our bodies, and all the energy that makes us go, are based on organic molecules where chains of carbon atoms serve as the backbone. The term “hydrate” refers to putting water in something.
So, in the most basic chemical sense, carbohydrates are chains of carbons (with oxygen and hydrogen) that form when water molecules are subtracted out.
The most basic units of all complex carbohydrates are simple sugars. These are short chains of carbon (usually with five or six carbon atoms) that often form rings, which tends to make them more stable.
Don’t worry, the chemistry torture won’t last long but you really do need a little chemistry introduction to carbohydrates in order to understand how they are digested in the body. Just think, there are actually people who choose chemistry as their major in college… truly, it’s a fascinating field if you don’t fight it too much.
Let’s look at the most basic chemical formula for carbohydrates:
(C + H2O)n
or you could write this as:
(CH2O)n
Now, in case that looks “greek” to you, let’s simplify it.
C = Carbon atom
H = Hydrogen atom
O = Oxygen atom
H2O = Water (one hydrogen atom bonded to two oxygen atoms; the looks of the molecule always reminds me of Micky Mouse!)
So, let’s go back to:
(C + H2O)n
or
(CH2O)n
This means that in almost all carbs (there are a few exceptions, such as the carbs you find in DNA and RNA, but we’ll ignore them), for every carbon atom you have, there are two hydrogen atoms and one oxygen atom, i.e. it’s a one to two ratio between carbon and hydrogen and a one to one ratio between carbon and oxygen.
Now look at those simplified formulas again and notice the n=6. This means that if a carb had six carbon atoms, there would be 12 hydrogen atoms, and six oxygen atoms. If a carb had 12 carbon atoms, there would be 24 hydrogen atoms and 12 oxygen atoms.
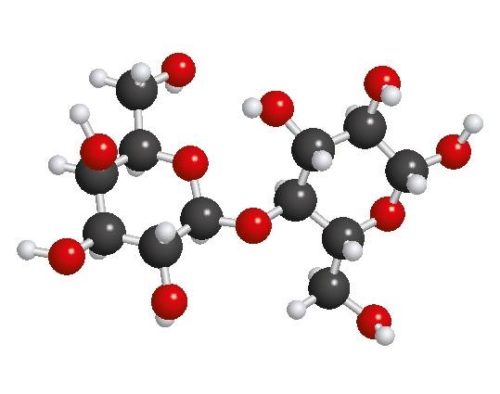
Guess what? The first example above, with six carbon atoms, is the chemical formula for glucose, one of the most important molecules in digestion. Glucose has six carbons, 12 hydrogens, and six oxygens. It is a type of simple carbohydrate, also called a simple sugar. Glucose is also the unit that most complex carbs you eat break down into and what fuels all your physiological needs.
Look at this graphical representation of the glucose molecule:
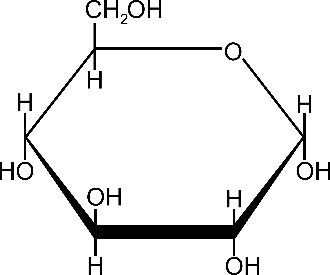
Now, here’s something you need to really pay attention to:
A COMPLEX CARB IS COMPOSED OF THOUSANDS OF SIMPLE CARBS (simple sugars like glucose) BONDED TOGETHER BY SPECIAL BONDS CALLED GLYCOSIDIC BONDS.
Look at this graphical representation of a glycosidic bond.
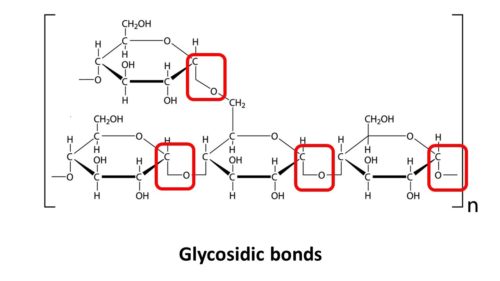
Don’t worry, we’re not going to give you a college level biochemistry lesson here. However, to understand carb digestion, you need to understand one more basic, but fundamental, point. It involves putting together complex carbs using the basic building blocks of simple sugars and then breaking them down again by breaking the bonds between those simple sugars.
Both of these processes involve WATER — H2O — at the level of the glycosidic bond.
To build complex carbs from simple sugar units, usually simple sugar rings, you have to SUBTRACT WATER. This is called condensation. Think about when you see water “condensation” on your windows in the early morning. Water has been REMOVED from the air and has collected on your windows.
To break down complex carbs, you have to ADD WATER. This is called HYDROLYSIS. Hydrolysis is the KEY to complex carb digestion, i.e. the key to how complex carbs are broken down. However, this chemical reaction MUST be facilitated by the right enzyme.
Hydrolysis is an interesting word. “Hydro” means water. “Lysis” means to split apart. “Hydrolysis” literally means to split apart with water. This “splitting apart” occurs right at the glycosidic bond.
All water-soluble complex carbohydrates are broken down until they form simple sugars that can be immediately utilized by your cells for energy or stored as glycogen. However, one thing we didn’t mention is that your liver, muscles, and other body parts that can store glycogen have a LIMITED storage capacity for glycogen. If you eat so many complex carbs that you surpass your immediate energy needs, and you surpass your limited storage room for glycogen, the rest will be converted to fat! Therefore, eating too many complex carbs can add just as much unwanted extra weight as eating too many simple sugars, i.e. simple carbs…. something to think about the next time you decide whether to supersize your fries or eat that bag of chips!

Leave a Reply
You must be logged in to post a comment.